|
Overall, the reaction takes place between a carboxylic acid and an acid anhydride to form an ester.Īspirin is not very soluble in water so the aspirin product will precipitate when water is added. Some of the other compounds, acetic anhydride and acetic acid, dissolve in water, but salicylic acid is only slightly soluble in cold water. Vacuum filtration will separate the crystalline aspirin away from everything else in the reaction mixture except for any salicylic acid that did not react. O0O- UF ChemDoodle Submit Answer Retry Entire Group 4 more group attempts This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. The excess acetic anhydride will be quenched (reacted) with the addition of water. 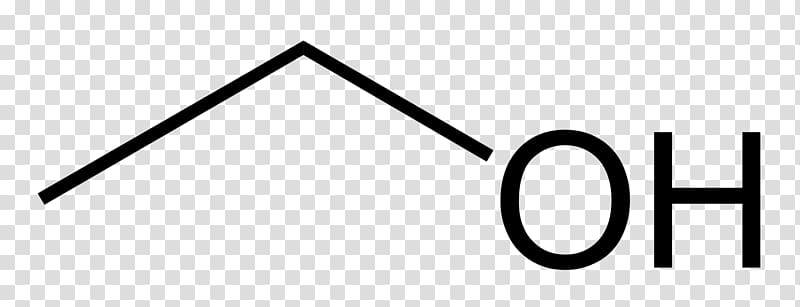
Here, n is the number of carbon atoms in the molecule. In this experiment, sulfuric acid will be used as the catalyst. Solution The general formula for alcohol The general formula for the alcohols is C n H 2 n + 1 OH. A small amount of a strong acid is used as a catalyst which speeds up the reaction. To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. The chemical formula of Isopropyl alcohol. molecular compound from its chemical formula. For unlimited access, please consider supporting the ChemDoodle team with a special offer for a ChemDoodle license. Hippocrates the ancient Greek physician, as well as Native Americans before Columbus’ time, prepared willow bark teas as headache remedies and other tonics. In modern times, salicylic acid is administered in the form of aspirin which is less irritating to the stomach than salicylic acid. The parent is heptane because the longest carbon chain contains. general formula for an alcohol chemdoodle. Ethers IUPAC nomenclature of ethers follows various guidelines. Its analgesic, antipyretic, and anti-inflammatory properties make it a powerful and effective drug to relive symptoms of pain, fever, and inflammation. Salicylic acid, whose name comes from Salix, the willow family of plants, was derived from willow bark extracts. For example, CH3OCH3 is named as Dimethyl ether. Separate the aspirin from the reaction by-products using vacuum filtration.Īnalyze the aspirin and estimate its purity.Īcetylsalicylic acid, commonly known as aspirin, is the most widely used drug in the world today. Open the Set Label dropdown menu and select H. Click the new carbon atom to change it to an sulfur atom.  
Open the dropdown menu next to the element button, and select S. Answer: Click, and click the carbon atom. \)Ĭonduct a chemical reaction to produce aspirin. Note The question begins with the ChemDoodle Sketcher tool displaying a single carbon atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
